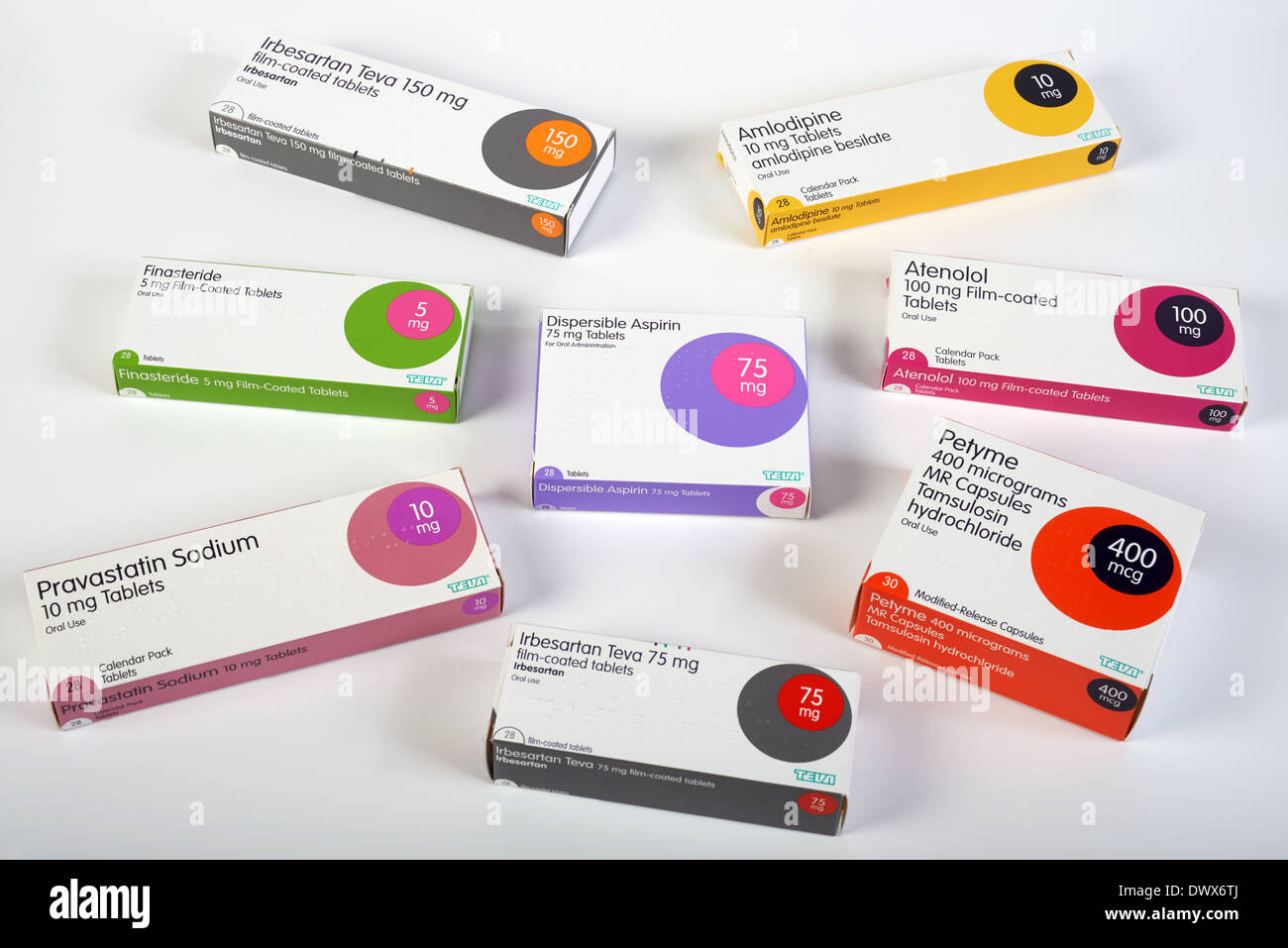
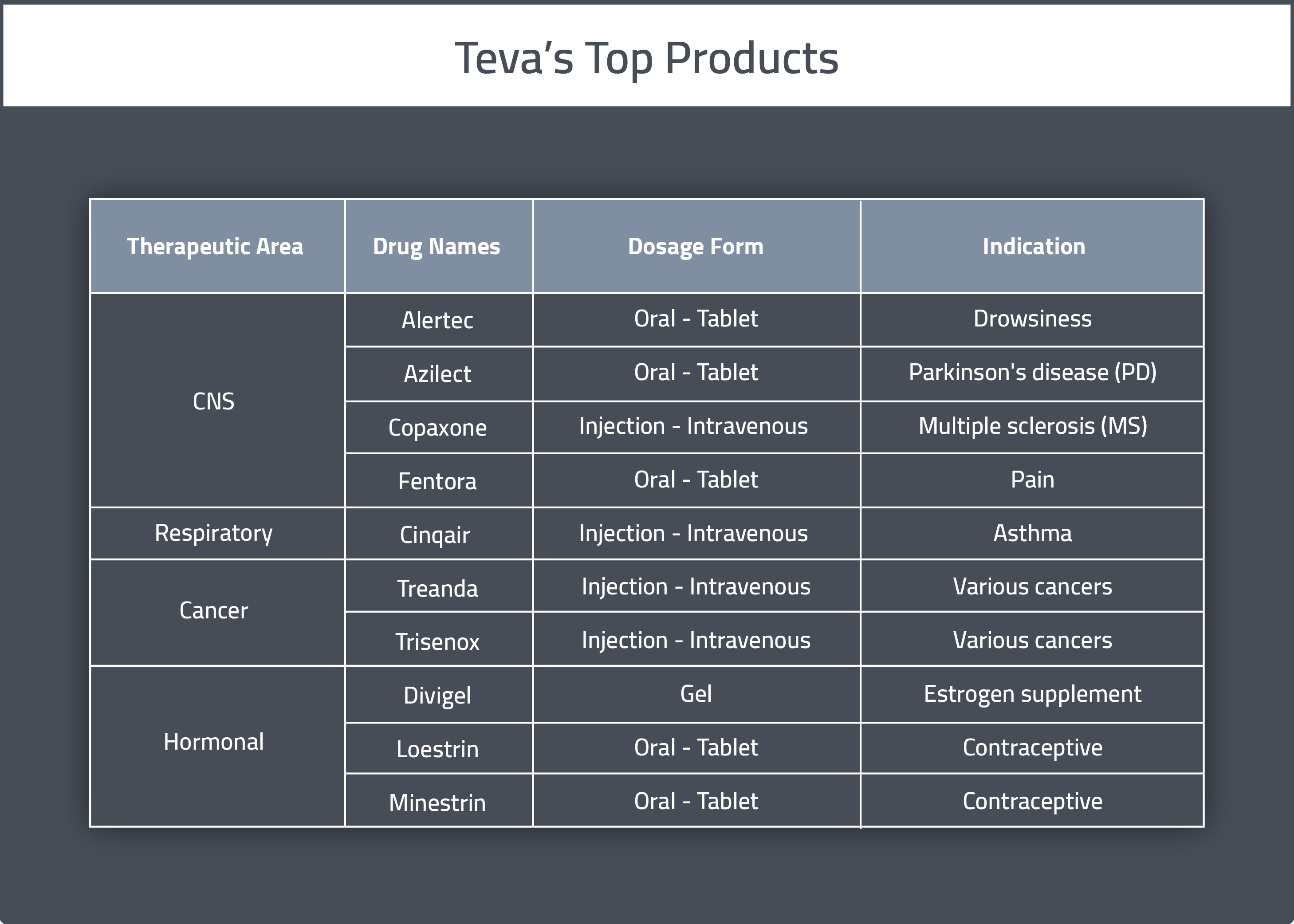
Teva completes $703M sale of women's health products to CVC Capital Partners. - Philadelphia Business Journal

Teva Pharmaceuticals USA Issues Voluntary Nationwide Recall of all Amlodipine/Valsartan Combination Tablets and Amlodipine/Valsartan/Hydrochlorothiazide Combination Tablets that are Within Expiry | FDA

Teva Pharmaceuticals - Next generation supply chain transformation by digitalisation at Teva by iThink Media - Issuu
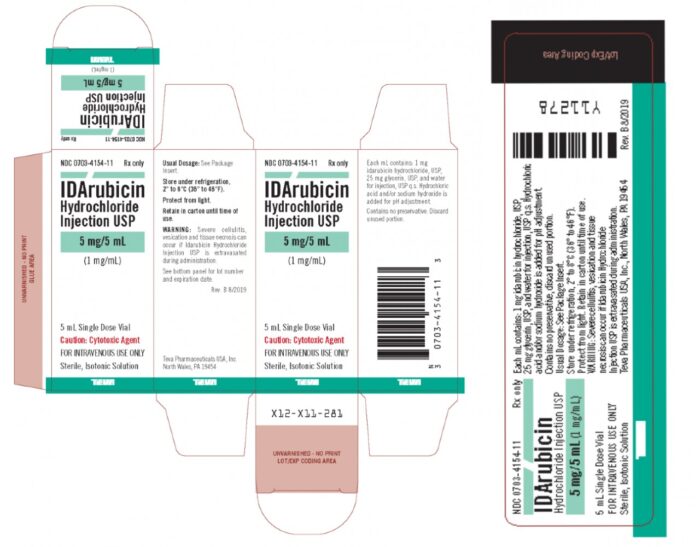
Teva Pharmaceuticals Recalls Acute Myeloid Leukemia Drug Over Particulate Matter Contamination - Top Class Actions